Clinical Studies
SPRAVATO™
Esketamine Therapy
CLINICAL TRIALS
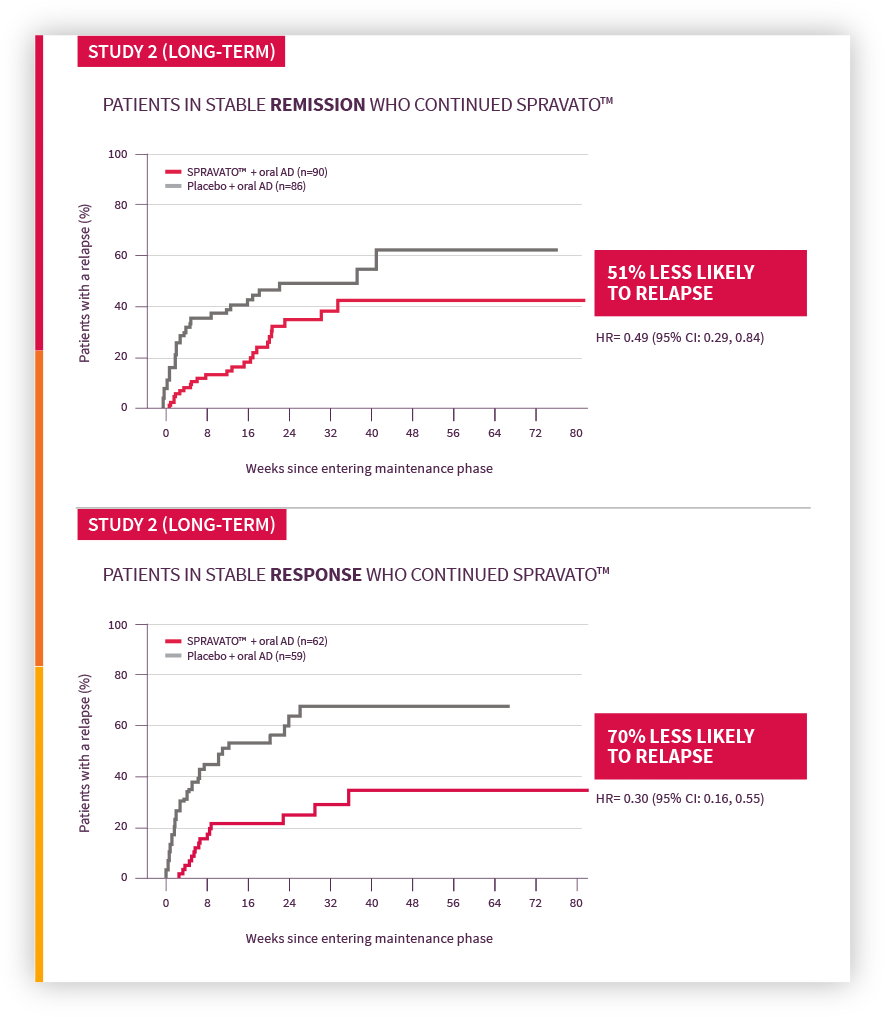
SPRAVATO™ has been studied in adults with treatment-resistant depression as an addition to an oral antidepressant regiment. Patients taking SPRAVATO™ experienced greater reduction of depression symptoms at four weeks (compared to those who received a placebo and oral antidepressant). After 16 weeks of therapy, those patients who stayed on SPRAVATO™ did better than those who stopped therapy.

Superior improvement in depression symptoms vs oral antidepressant (AD) plus placebo at Week 4 (1,2).
1. SPRAVATO™ [Prescribing Information]. Titusville, NJ: Janssen Pharmaceuticals, Inc. March 2019.
2. Popova V, Daly EJ, Trivedi M, et al. Randomized, double-blind study of flexibly-dosed intranasal esketamine plus oral antidepressant vs. active control in treatment-resistant depression. Presented at: 2018 Annual Meeting of the American Society of Clinical Psychopharmacology (ASCP); May 29-June 1, 2018; Miami, FL.
